Top Suppliers Dry Heat Sterilizer Validation - Single-Channel Mechanical Pipettor – Bioendo
Top Suppliers Dry Heat Sterilizer Validation - Single-Channel Mechanical Pipettor – Bioendo Detail:
Single-Channel Mechanical Pipettor
1. Product Information
Single channel mechanical pipette is ideal tool to support endotoxin detection with Lyophilized Amebocyte Lysate which covers gel-clot technique, kinetic turbidimetric technique, kinetic chromogenic technique, and end-point chromogenic technique. All pipettors are produced by following ISO8655 – 2:2002. The quality control involves gravimetric testing of each pipette with distilled water at 22℃.
2. Product Features:
- Light weight, economic, low force design
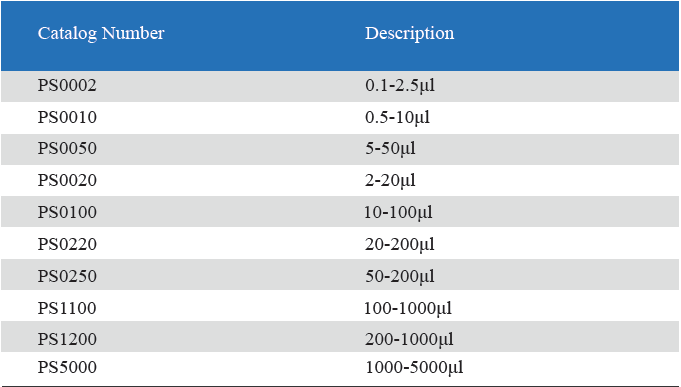
- Measuring volume range from 0.1μL to 5mL
- Easy to calibrate and maintain with tool supplied
- Design helps to avoid repetitive strain injuries
- Calibrated in accordance with ISO8655. Each pipettor supplied with individual test certificate
- The low part is available for autoclaving
Product detail pictures:

Related Product Guide:
We attempt for excellence, company the customers", hopes to be the top cooperation team and dominator company for personnel, suppliers and customers, realizes price share and continual marketing for Top Suppliers Dry Heat Sterilizer Validation - Single-Channel Mechanical Pipettor – Bioendo , The product will supply to all over the world, such as: Norway, Uruguay, Boston, We are very responsible for all details on our customers order no matter on warranty quality, satisfied prices, quick delivery, on time communication, satisfied packing, easy payment terms, best shipment terms, after sales service etc. We provide one-stop service and best reliability to our every customers. We work hard with our customers, colleagues, workers to make a better future.
It is a very good, very rare business partners, looking forward to the next more perfect cooperation!








