OEM Manufacturer BET Test Procedure - Mini Dry Heat Incubator – Bioendo
OEM Manufacturer BET Test Procedure - Mini Dry Heat Incubator – Bioendo Detail:
Dry heat incubator single module
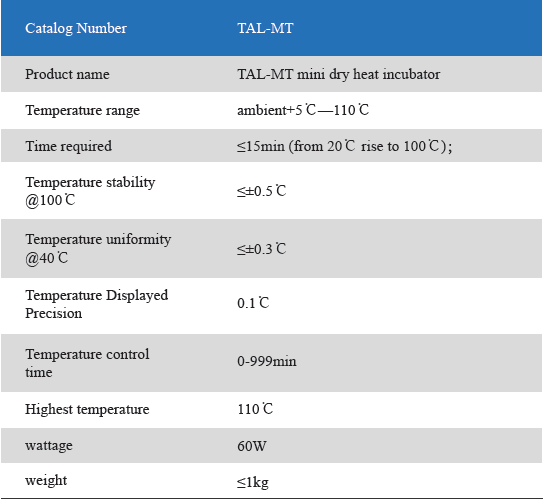
1. Product information
The Mini Dry Heat Incubator is a micro-processor controlled heating block with semi conductor heating technology.It adapts onboard use, smart, light and convenient for movement, suit for any kind of occasions. Especially good for the incubation of the gel clot LAL assay, LAL chromogenic endpoint assay incubation.
2. Product features
1. Unique designed. Smart and light, convenient movement, suit for various occasions.
2. LCD simultaneously display setting and actual time and temperature.Temperature calibration function.
3. Automatic fault detection function with buzzer alarm.
4. 24V DC input power, built-in over-temperature protection device.
5. Various of blocks for optional choice. Convenient for replacement. Easy cleaning and disinfection.
Product detail pictures:



Related Product Guide:
Our improvement depends on the superior equipment, excellent talents and continuously strengthened technology forces for OEM Manufacturer BET Test Procedure - Mini Dry Heat Incubator – Bioendo , The product will supply to all over the world, such as: Sevilla, Panama, Malta, Our products are mainly exported to Southeast Asia, the Middle East, North America and Europe. Our quality is surely guaranteed. If you are interested in any of our products or would like to discuss a custom order, please feel free to contact us. We are looking forward to forming successful business relationships with new clients around the world in the near future.
The factory has advanced equipment, experienced staffs and good management level, so product quality had assurance, this cooperation is very relaxed and happy!






