Manufacturing Companies for Advantages Of LAL Test - Eight-channel Mechanical Pipette – Bioendo
Manufacturing Companies for Advantages Of LAL Test - Eight-channel Mechanical Pipette – Bioendo Detail:
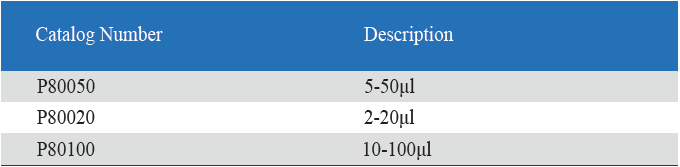
Eight-Channel Mechanical Pipettor
1. Product information
All multi-channel mechanical pipettor have been quality tested according to ISO8655-2:2002 with calibration certificate. The quality control involves gravimetric testing of each pipette with distilled water at 22℃. The multichannel mechanical pipettor is idea for the detectionof bacterial endotoxin lal endotoxin testing by kinetic turbidimetric andkinetic chromogenic method.
- Eight-Channel Mechanical Pipettor is available for standard 96-well plate
- Dispensing head rotates for optimum pipetting convenience
- Individual piston and tip cone assemblies allow easy repair and maintenance
- Compound material tip cone design allows visual seal verification
- Can be used with universal style pipette tips
-good for kinetic chromogenic, kinetic turbidimetric TAL orend-point chromogenic TAL endotoxin assay
Product detail pictures:
Related Product Guide:
With our leading technology also as our spirit of innovation,mutual cooperation, benefits and development, we are going to build a prosperous future jointly with your esteemed company for Manufacturing Companies for Advantages Of LAL Test - Eight-channel Mechanical Pipette – Bioendo , The product will supply to all over the world, such as: Belize, Venezuela, Guinea, Only for accomplishing the good-quality product to meet customer's demand, all of our products have been strictly inspected before shipment. We always think about the question on the side of the customers, because you win, we win!
High Quality, High Efficiency, Creative and Integrity, worth having long-term cooperation! Looking forward to the future cooperation!








