Special Design for Bacterial Endotoxin Test Methods - Depyrogenated Sample Bottles ( Depyrogenated Galssware ) – Bioendo
Special Design for Bacterial Endotoxin Test Methods - Depyrogenated Sample Bottles ( Depyrogenated Galssware ) – Bioendo Detail:
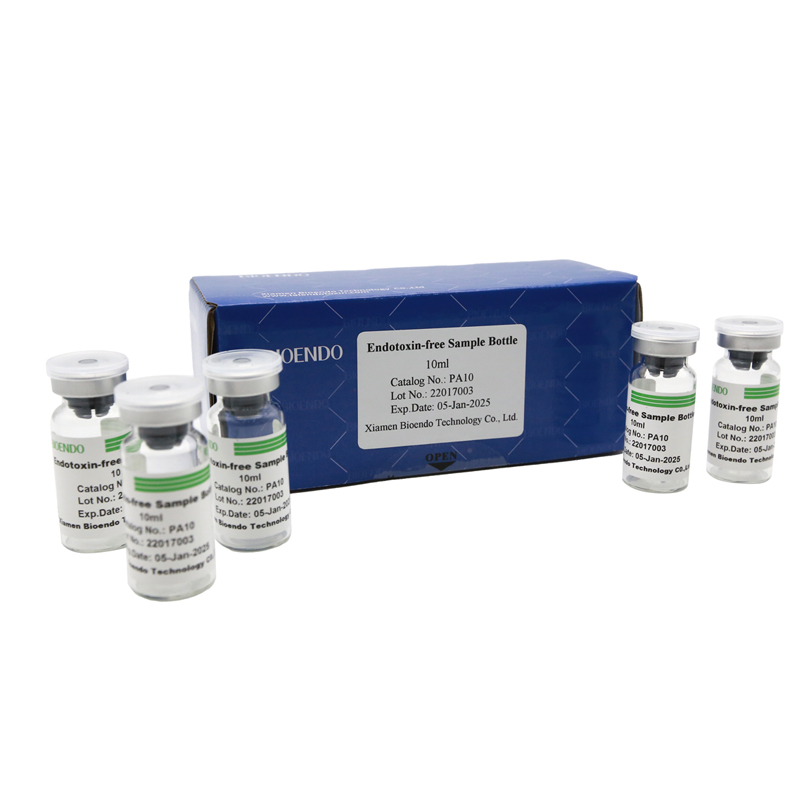
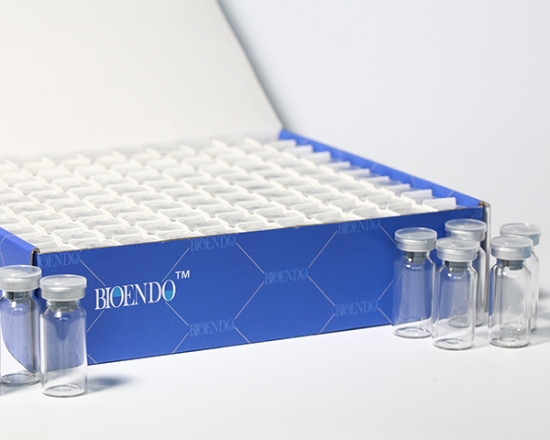
Depyrogenated Sample Bottle
1. Product information
We offer various of low endotoxin, pyrogen free accessories products,includes Water for Bacterial Endotoxins Test, pyrogen-free test tubes, pyrogen-free pipette tips, pyrogen free microplates and sample bottles for your conveniences. Amoung the sample bottle have two types, one is depyrogenated glassware and the other is depyrogenated plasticware, both endotoxin free level. High quality depyrogenated low endotoxin pyrogen free products insure the success of your experiments.
Depyrogenated (Endotoxin Free) Sample container(endotoxin free bottle, pyrogen free bottle, pyrogen free sample bottle)are glass bottles contain less than 0.005 EU/ml endotoxin. These bottles could be used to store various samples for lps endotoxin test, such as protein solution, vaccines, DNA solutions, dialysate, water for injections, etc., for endotoxin testing. Come with endotoxin free seals.
2. Product parameter
Endotoxin level < 0.005 EU/ml
3. Product features and application
For preparing and storage of the test samples.
| Catalog Number | Descriptions | Package |
| PA2 | Endotoxin free sample glass ampoule, 2ml | 10Pcs/Pack |
| PA10 | Endotoxin free sample glass vial, 10ml | 10Pcs/Pack or 110Pcs/Pack |
| PA50 | Endotoxin free sample glass vial, 50ml | 10Pcs/Pack |
| PA125 | Pyrogen free sample bottle, 125ml | 1Pcs/Pack |
| PA500 | Pyrogen free sample bottle, 500ml | 1Pcs/Pack |
Product detail pictures:





Related Product Guide:
owing to excellent assistance, a variety of top of the range items, aggressive costs and efficient delivery, we take pleasure in a very good standing amongst our shoppers. We've been an energetic corporation with wide market for Special Design for Bacterial Endotoxin Test Methods - Depyrogenated Sample Bottles ( Depyrogenated Galssware ) – Bioendo , The product will supply to all over the world, such as: Bangladesh, Manchester, Angola, We have a large share in global market. Our company has strong economic strength and offers excellent sale service. We have established faith, friendly, harmonious business relationship with customers in different countries. , such as Indonesia, Myanmar, Indi and other Southeast Asian countries and European, African and Latin American countries.
The after-sale warranty service is timely and thoughtful, encounter problems can be resolved very quickly, we feel reliable and secure.