OEM/ODM Supplier Kit LAL - Single-Channel Mechanical Pipettes (Semi-sterile) – Bioendo
OEM/ODM Supplier Kit LAL - Single-Channel Mechanical Pipettes (Semi-sterile) – Bioendo Detail:
Single-Channel Mechanical Pipettes (Semi-sterile)
1. Product Information
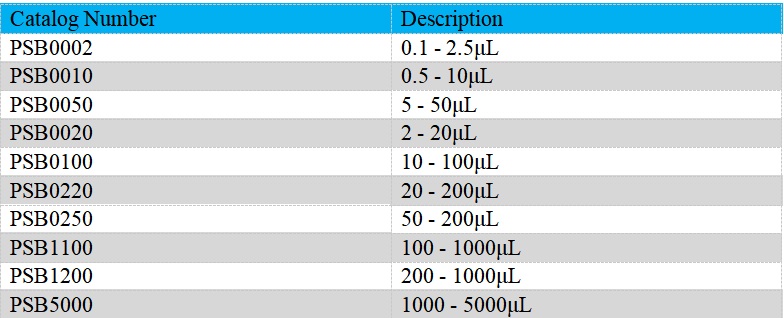
The Single-Channel Mechanical Pipettes (Semi-sterile) is adjustable, which could be used to dispense liquid precisely. Measuring volume of our Single-Channel Mechanical Pipettes (Semi-sterile) ranges from 0.1μL to 5mL. Products are produced based on ISO8655/DIN12650. It could be widely employed on endotoxin detection, etc..
2. Product Features
- Light weight, economic
- The pipettes cover volume range from 0.1μL to 5mL
- Semi-sterile
- Easy calibration and maintenance
Product detail pictures:

Related Product Guide:
In an effort to provide you advantage and enlarge our business enterprise, we even have inspectors in QC Staff and assure you our greatest provider and item for OEM/ODM Supplier Kit LAL - Single-Channel Mechanical Pipettes (Semi-sterile) – Bioendo , The product will supply to all over the world, such as: Croatia, Sudan, Johannesburg, Our company insists on the principle of "Quality First, Sustainable Development", and takes "Honest Business, Mutual Benefits" as our developable goal. All members sincerely thank all old and new customers' support. We will keep working hard and offering you the highest-quality products and service.
With a positive attitude of "regard the market, regard the custom, regard the science", the company works actively to do research and development. Hope we have a future business relationships and achieving mutual success.






