OEM/ODM China Kinetic Turbidimetric Method of endotoxin testing - Kinetic Turbidimetric Amebocyte Lysate Vial – Bioendo
OEM/ODM China Kinetic Turbidimetric Method of endotoxin testing - Kinetic Turbidimetric Amebocyte Lysate Vial – Bioendo Detail:
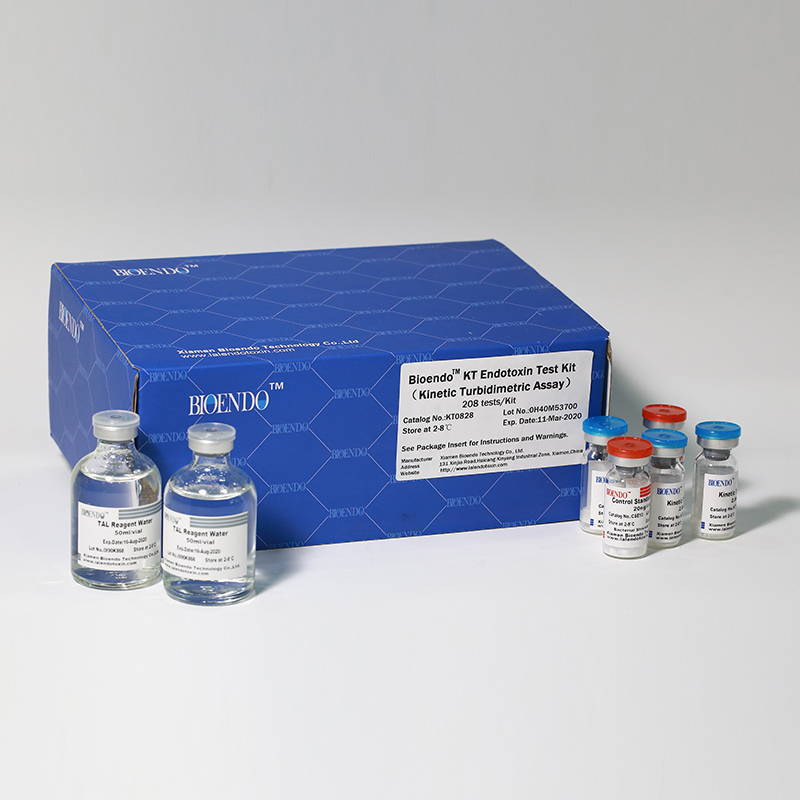
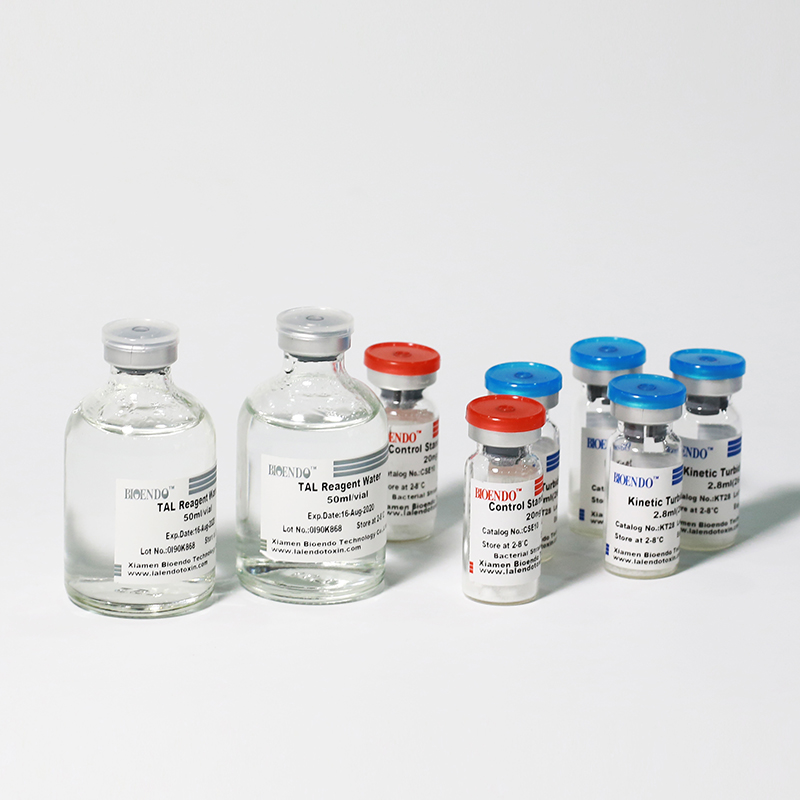
Kinetic Turbidimetric Amebocyte Lysate Vial
1. Product Introduction
Kinetic Turbidimetric Amebocyte Lysate Vial is developed based on the principle that the time needed to reach a certain absorbance increase (onset OD), i.e. onset time, is negatively correlated with the endotoxin concentration. Sensitivity could reach 0.005EU/ml, and the detection could reach four orders of magnitude. It is specially suitable for pharmaceuticals industry to monitor endotoxin concentration.
2. Product Parameter:
Assay range:0.005-50EU/ml; 0.01 – 10EU/ml
3. Product Application
End-product endotoxin (pyrogen) qualification, Water for injection endotoxin assay, raw material endotoxin testing or endotoxin level monitoring during manufacturing process for pharmaceutical companies or medical devices manufacturers.
Note:
Lyophilized Amebocyte Lysate reagent manufactured by Bioendo is made from amebocyte lysate from the horseshoe crab (Tachypleus tridentatus).
|
Catalog No. |
ml/vial |
Tests/Vial |
Vials/Pack |
Sensitivity EU/ml |
|
KT17 |
1.7 |
16 |
10 |
0.01-10EU/ml |
|
KT17S |
1.7 |
16 |
10 |
0.005-5EU/ml, 0.01-10EU/ml |
|
KT52 |
5.2 |
50 |
10 |
0.01-10EU/ml |
|
KT52S |
5.2 |
50 |
10 |
0.005-5EU/ml, 0.01-10EU/ml |
The Lyophilized Amebocyte Lysate reagent sensitivity and the Control Standard Endotoxin potency are assayedagainst USP Reference Standard Endotoxin. The Lyophilized Amebocyte Lysate reagent kits come withproduct instruction, Certificate of Analysis.


Product detail pictures:

Related Product Guide:
We normally follow the basic principle "Quality Initial, Prestige Supreme". We've been fully committed to offering our consumers with competitively priced good quality merchandise, prompt delivery and professional support for OEM/ODM China Kinetic Turbidimetric Method of endotoxin testing - Kinetic Turbidimetric Amebocyte Lysate Vial – Bioendo , The product will supply to all over the world, such as: Cape Town, Bangladesh, Greek, Our qualified engineering team will usually be prepared to serve you for consultation and feedback. We are able to also deliver you with absolutely free samples to meet your needs. Best efforts might be made to offer you the ideal service and products. For anyone who is interested in our company and items, please make contact with us by sending us emails or contact us right away. In order to know our solutions and organization. ar more, you can come to our factory to determine it. We are going to usually welcome guests from around the globe to our corporation. o create small business relations with us. Please really feel no cost to speak to us for enterprise. nd we believe we are going to share the most effective trading practical experience with all our merchants.
The company comply with the contract strict, a very reputable manufacturers, worthy a long-term cooperation.