OEM/ODM China Dry heat sterilizer validation by ECV - Eight-channel Mechanical Pipette – Bioendo
OEM/ODM China Dry heat sterilizer validation by ECV - Eight-channel Mechanical Pipette – Bioendo Detail:
Eight-Channel Mechanical Pipettor
1. Product information
All multi-channel mechanical pipettor have been quality tested according to ISO8655-2:2002 with calibration certificate. The quality control involves gravimetric testing of each pipette with distilled water at 22℃. The multichannel mechanical pipettor is idea for the detectionof bacterial endotoxin lal endotoxin testing by kinetic turbidimetric andkinetic chromogenic method.
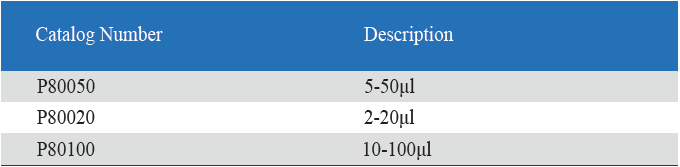
- Eight-Channel Mechanical Pipettor is available for standard 96-well plate
- Dispensing head rotates for optimum pipetting convenience
- Individual piston and tip cone assemblies allow easy repair and maintenance
- Compound material tip cone design allows visual seal verification
- Can be used with universal style pipette tips
-good for kinetic chromogenic, kinetic turbidimetric TAL orend-point chromogenic TAL endotoxin assay
Product detail pictures:

Related Product Guide:
Quality First,and Customer Supreme is our guideline to provide the best service to our customers.Nowadays, we are trying our best to become one of the best exporters in our field to meet customers more need for OEM/ODM China Dry heat sterilizer validation by ECV - Eight-channel Mechanical Pipette – Bioendo , The product will supply to all over the world, such as: Southampton, Cancun, Casablanca, They're durable modeling and promoting well all over the world. Under no circumstances disappearing key functions in a brief time, it's a should for you personally of fantastic quality. Guided by the principle of Prudence, Efficiency, Union and Innovation. the business make an awesome efforts to expand its international trade, raise its enterprise. rofit and improve its export scale. We are confident that we'll have a vibrant prospect and to be distributed all over the world in the years to come.
This enterprise in the industry is strong and competitive, advancing with the times and develop sustainable, we are very pleased to have a opportunity to cooperate!