OEM/ODM China Bacterial Endotoxin Test Gel Clot Method Sop - Bioendo GC Endotoxin Test Kit (Gel Clot Assay) – Bioendo
OEM/ODM China Bacterial Endotoxin Test Gel Clot Method Sop - Bioendo GC Endotoxin Test Kit (Gel Clot Assay) – Bioendo Detail:
Bioendo GC Endotoxin Test Kit
Bioendo GC Endotoxin Test Kit (Gel Clot Assay)
1. Product Information
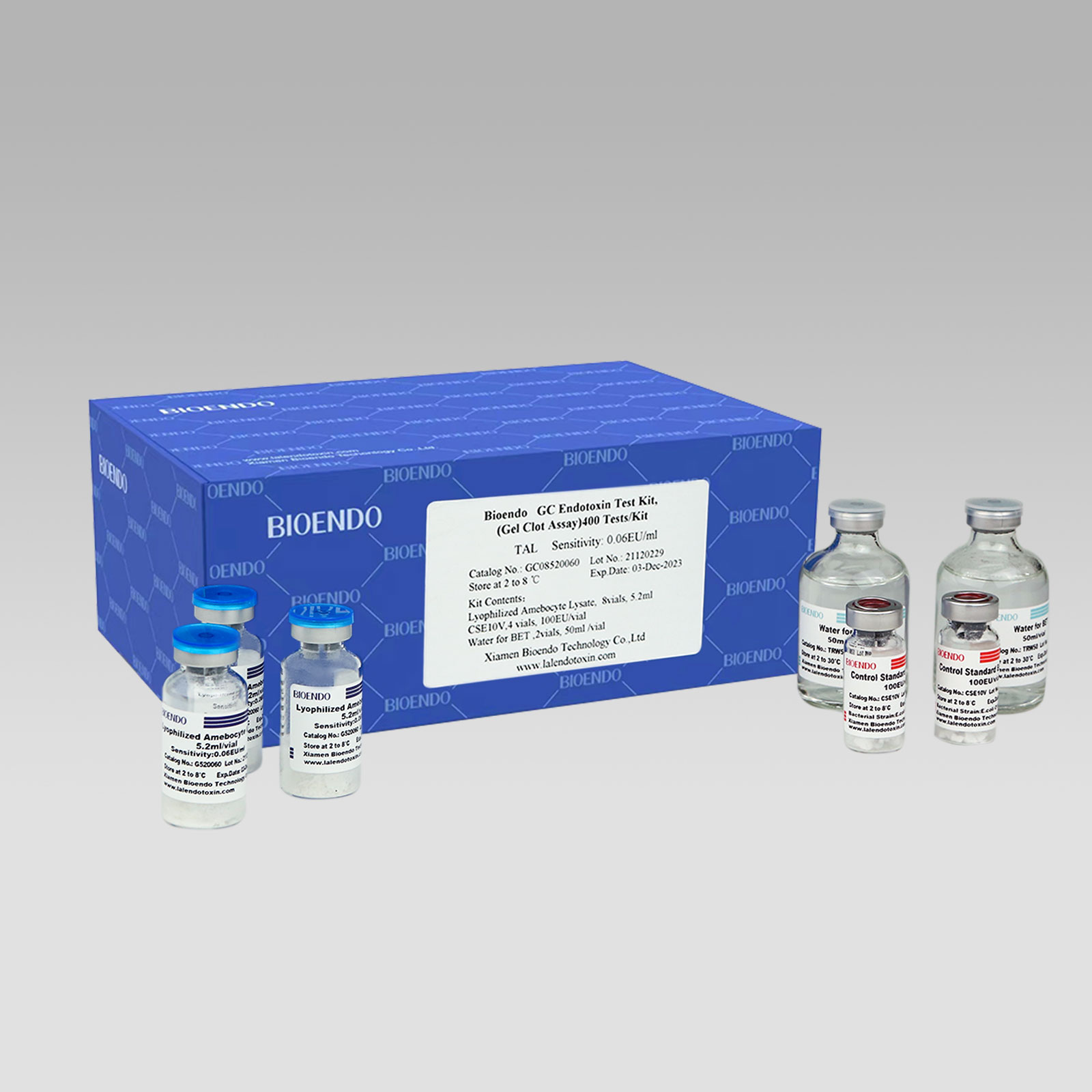
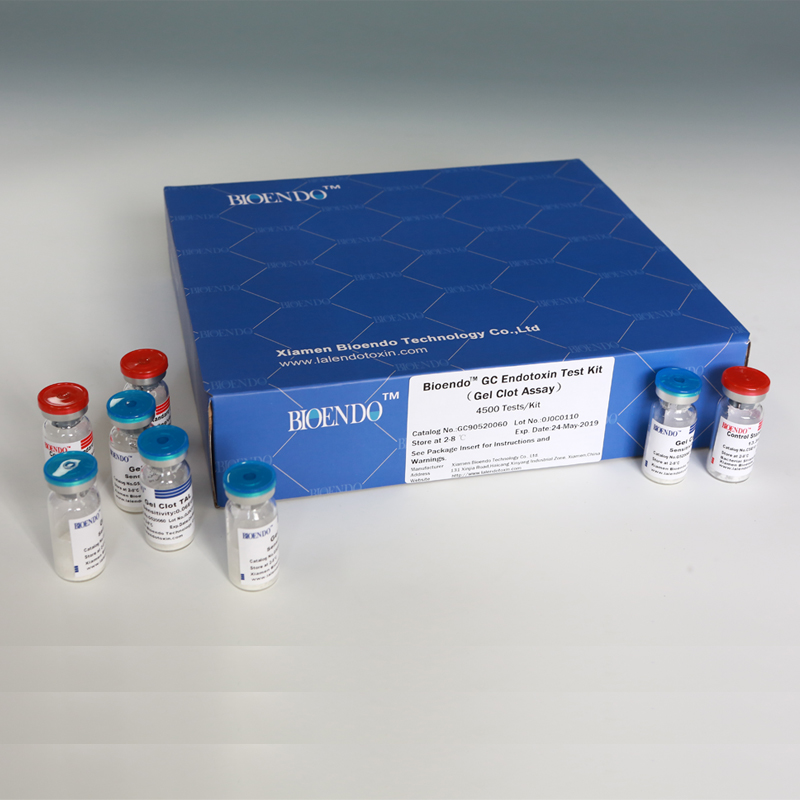
Bioendo GC Endotoxin Test Kit (Gel Clot Assay) contains 128 Tests/Kit, 400Tests/Kit, 1600Tests/Kit, and 4500 Tests/Kit. And sensitivity of Amebocyte Lysate included by the Kit is 0.03 EU/ml, 0.06 EU/ml, 0.125 EU/ml, 0.25 EU/ml, 0.5 EU/ml. The Kit contains Amebocyte Lysate, CSE and Water for BET. Four kinds of configuration of gel-clot endotoxin assay kit to satisfied the demand. This means you could choose different configuration kits according to the requirement.
2. Product Parameter
Sensitivities: 0.03EU/ml, 0.06EU/ml, 0.125EU/ml, 0.25EU/ml, 0.5 EU/ml
3.Product Feature and Application
Full range of sensitivities to meet the demand. No special instrument is needed for endotoxin detection with Bioendo GC Endotoxin Test Kit (Gel Clot Assay). It is the best choice when you need to do lots of endotoxin detection.
Note:
Lyophilized Amebocyte Lysate (LAL/TAL) reagent manufactured by Bioendo is made from amebocyte lysate derived blood of horseshoe crab.
|
Catalog No. |
Sensitivity (EU/ml) |
Description |
Kit Contents |
|
GC90520030 |
0.03 |
Bioendo™ GC Endotoxin Test Kit (Gel Clot Assay) 4500 Tests/Kit |
90 Gel Clot Lyophilized Amebocyte Lysate, 5.2ml (50 Tests/Vial); 20 CSE10V; |
|
GC90520060 |
0.06 |
||
|
GC90520125 |
0.125 |
||
|
GC90520250 |
0.25 |
||
|
GC90520500 |
0.5 |
||
|
GC08520030 |
0.03 |
Bioendo™ GC Endotoxin Test Kit (Gel Clot Assay) 400 Tests/Kit |
8 Gel Clot Lyophilized Amebocyte Lysate, 5.2ml (50 Tests/Vial); 4 CSE10V; 2 Water for BET, 50ml/vial; |
|
GC08520060 |
0.06 |
||
|
GC08520125 |
0.125 |
||
|
GC08520250 |
0.25 |
||
|
GC08520500 |
0.5 |
||
|
GC100170030 |
0.03 |
Bioendo™ GC Endotoxin Test Kit (Gel Clot Assay) 1600 Tests/Kit |
100 Gel Clot Lyophilized Amebocyte Lysate, 1.7ml (16 Tests/Vial); 10 CSE10V; |
|
GC100170060 |
0.06 |
||
|
GC100170125 |
0.125 |
||
|
GC100170250 |
0.25 |
||
|
GC100170500 |
0.5 |
||
|
GC08170030 |
0.03 |
Bioendo™ GC Endotoxin Test Kit (Gel Clot Assay) 128 Tests/Kit |
8 Gel Clot Lyophilized Amebocyte Lysate, 1.7ml (16 Tests/Vial); 4 CSE10V; 2 Water for BET, 50ml/vial; |
|
GC08170060 |
0.06 |
||
|
GC08170125 |
0.125 |
||
|
GC08170250 |
0.25 |
||
|
GC08170500 |
0.5 |
Product condition:
Endotoxin Test kit’s sensitivity and the Control Standard Endotoxin potency are assayed against USP Reference Standard Endotoxin. The Endotoxin Test reagentkits come with product instruction, Certificate of Analysis.
Product detail pictures:

Related Product Guide:
We purpose to understand high quality disfigurement with the output and supply the top service to domestic and overseas buyers wholeheartedly for OEM/ODM China Bacterial Endotoxin Test Gel Clot Method Sop - Bioendo GC Endotoxin Test Kit (Gel Clot Assay) – Bioendo , The product will supply to all over the world, such as: Algeria, Brasilia, Turkey, Our company follows laws and international practice. We promise to be responsible for friends, customers and all partners. We would like to establish a long-term relationship and friendship with every customer from all over the world on the basis of mutual benefits. We warmly welcome all old and new customers to visit our company to negotiate business.
Speaking of this cooperation with the Chinese manufacturer, I just want to say"well dodne", we are very satisfied.