OEM Manufacturer BET Test Procedure - Mini Dry Heat Incubator – Bioendo
OEM Manufacturer BET Test Procedure - Mini Dry Heat Incubator – Bioendo Detail:
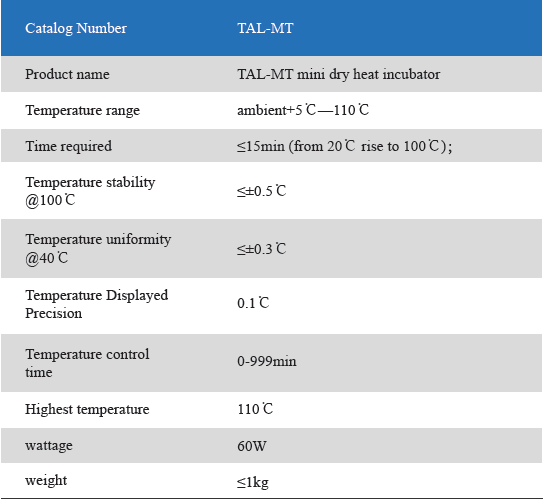
Dry heat incubator single module
1. Product information
The Mini Dry Heat Incubator is a micro-processor controlled heating block with semi conductor heating technology.It adapts onboard use, smart, light and convenient for movement, suit for any kind of occasions. Especially good for the incubation of the gel clot LAL assay, LAL chromogenic endpoint assay incubation.
2. Product features
1. Unique designed. Smart and light, convenient movement, suit for various occasions.
2. LCD simultaneously display setting and actual time and temperature.Temperature calibration function.
3. Automatic fault detection function with buzzer alarm.
4. 24V DC input power, built-in over-temperature protection device.
5. Various of blocks for optional choice. Convenient for replacement. Easy cleaning and disinfection.
Product detail pictures:



Related Product Guide:
Our objective is to present premium quality products at aggressive prices, and top-notch services to buyers around the world. We have been ISO9001, CE, and GS certified and strictly adhere to their excellent specifications for OEM Manufacturer BET Test Procedure - Mini Dry Heat Incubator – Bioendo , The product will supply to all over the world, such as: Nigeria, New Orleans, Southampton, Our company insists on the purpose of "takes service priority for standard, quality guarantee for the brand, do business in good faith, to offer skilled, rapid, accurate and timely service for you". We welcome old and new customers to negotiate with us. We are going to serve you with all sincerity!
Adhering to the business principle of mutual benefits, we have a happy and successful transaction, we think we will be the best business partner.








