OEM Factory for BET Test Principle - Eight-channel Mechanical Pipette – Bioendo
OEM Factory for BET Test Principle - Eight-channel Mechanical Pipette – Bioendo Detail:
Eight-Channel Mechanical Pipettor
1. Product information
All multi-channel mechanical pipettor have been quality tested according to ISO8655-2:2002 with calibration certificate. The quality control involves gravimetric testing of each pipette with distilled water at 22℃. The multichannel mechanical pipettor is idea for the detectionof bacterial endotoxin lal endotoxin testing by kinetic turbidimetric andkinetic chromogenic method.
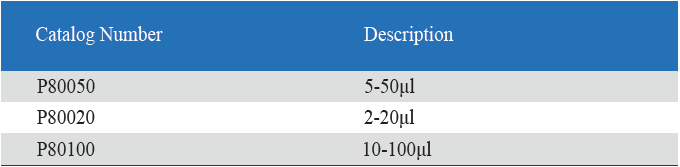
- Eight-Channel Mechanical Pipettor is available for standard 96-well plate
- Dispensing head rotates for optimum pipetting convenience
- Individual piston and tip cone assemblies allow easy repair and maintenance
- Compound material tip cone design allows visual seal verification
- Can be used with universal style pipette tips
-good for kinetic chromogenic, kinetic turbidimetric TAL orend-point chromogenic TAL endotoxin assay
Product detail pictures:

Related Product Guide:
With a sound business credit, excellent after-sales service and modern manufacturing facilities, we have earned an excellent reputation among our customers across the world for OEM Factory for BET Test Principle - Eight-channel Mechanical Pipette – Bioendo , The product will supply to all over the world, such as: Hungary, Vancouver, Cyprus, Thus far, our item associated with printer dtg a4 may be shown in most foreign nations as well as urban centers, which are sought after simply by targeted traffic. We all highly imagine that now we have the full capacity to present you with contented merchandise. Desire to collect requests of your stuff and produce the long-term co-operation partnership. We very seriously promise:Csame top quality, better price; exact same selling price, higher quality.
Timely delivery, strict implementation of the contract provisions of the goods, encountered special circumstances, but also actively cooperate, a trustworthy company!