OEM Customized Endotixins - Endotoxin Assay and (1,3)-ß-D-glucan assay software – Bioendo
OEM Customized Endotixins - Endotoxin Assay and (1,3)-ß-D-glucan assay software – Bioendo Detail:
Endotoxin and (1,3)-ß-D-glucan assay software
1. Product information
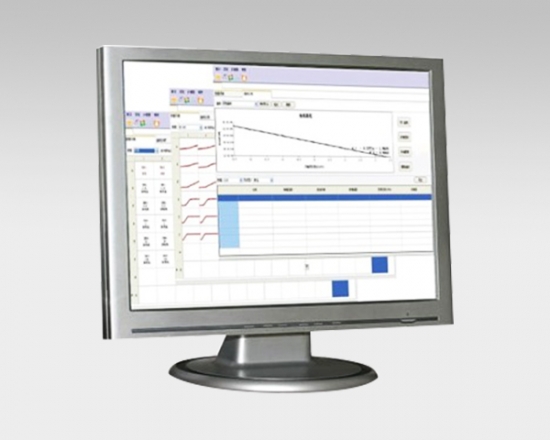
Endotoxin and (1,3)-ß-D-glucan assay software is a powerful kinetic data analysis software,which gives the user data acquisition and processing of the maximum flexibility.
Features:
• Apply to Endotoxin assay,(1,3)-ß-D-glucan assay and ELISA data analysis
• With standard version and clinical diagnostic version for the different user groups.
• Data could be output transitions and connected to the LIS system.
• Customizable endotoxin test reports.
• Data analyzed by onset time, average rate, maximum rate and other methods.
• Data linear fitting or polynomial fitting.
• Real-time backup of the original data read.
• Integration of variety of kinetic microplate readers.

Product detail pictures:

Related Product Guide:
We now have a highly efficient crew to deal with inquiries from clients. Our intention is "100% shopper pleasure by our merchandise quality, price tag & our staff service" and take pleasure in a very good standing amongst purchasers. With quite a few factories, we can easily provide a wide vary of OEM Customized Endotixins - Endotoxin Assay and (1,3)-ß-D-glucan assay software – Bioendo , The product will supply to all over the world, such as: kazakhstan, Iran, Germany, Besides strong technical strength, we also introduce advanced equipment for inspection and conduct strict management. All the staff of our company welcome friends both at home and abroad to come for visits and business on the basis of equality and mutual benefit. If you are interested in any of our items, please feel free to contact us for quotation and product details.
In general, we are satisfied with all aspects, cheap, high-quality, fast delivery and good procuct style, we will have follow-up cooperation!