Low price for Bacterial Endotoxin Test Kinetic Turbidimetric Method – Kinetic Turbidimetric Amebocyte Lysate Vial – Bioendo
Low price for Bacterial Endotoxin Test Kinetic Turbidimetric Method – Kinetic Turbidimetric Amebocyte Lysate Vial – Bioendo Detail:
Kinetic Turbidimetric Amebocyte Lysate Vial
1. Product Introduction
Kinetic Turbidimetric Amebocyte Lysate Vial is developed based on the principle that the time needed to reach a certain absorbance increase (onset OD), i.e. onset time, is negatively correlated with the endotoxin concentration. Sensitivity could reach 0.005EU/ml, and the detection could reach four orders of magnitude. It is specially suitable for pharmaceuticals industry to monitor endotoxin concentration.
2. Product Parameter:
Assay range:0.005-50EU/ml; 0.01 – 10EU/ml
3. Product Application
End-product endotoxin (pyrogen) qualification, Water for injection endotoxin assay, raw material endotoxin testing or endotoxin level monitoring during manufacturing process for pharmaceutical companies or medical devices manufacturers.
Note:
Lyophilized Amebocyte Lysate reagent manufactured by Bioendo is made from amebocyte lysate from the horseshoe crab (Tachypleus tridentatus).
|
Catalog No. |
ml/vial |
Tests/Vial |
Vials/Pack |
Sensitivity EU/ml |
|
KT17 |
1.7 |
16 |
10 |
0.01-10EU/ml |
|
KT17S |
1.7 |
16 |
10 |
0.005-5EU/ml, 0.01-10EU/ml |
|
KT52 |
5.2 |
50 |
10 |
0.01-10EU/ml |
|
KT52S |
5.2 |
50 |
10 |
0.005-5EU/ml, 0.01-10EU/ml |
The Lyophilized Amebocyte Lysate reagent sensitivity and the Control Standard Endotoxin potency are assayedagainst USP Reference Standard Endotoxin. The Lyophilized Amebocyte Lysate reagent kits come withproduct instruction, Certificate of Analysis.
Product detail pictures:
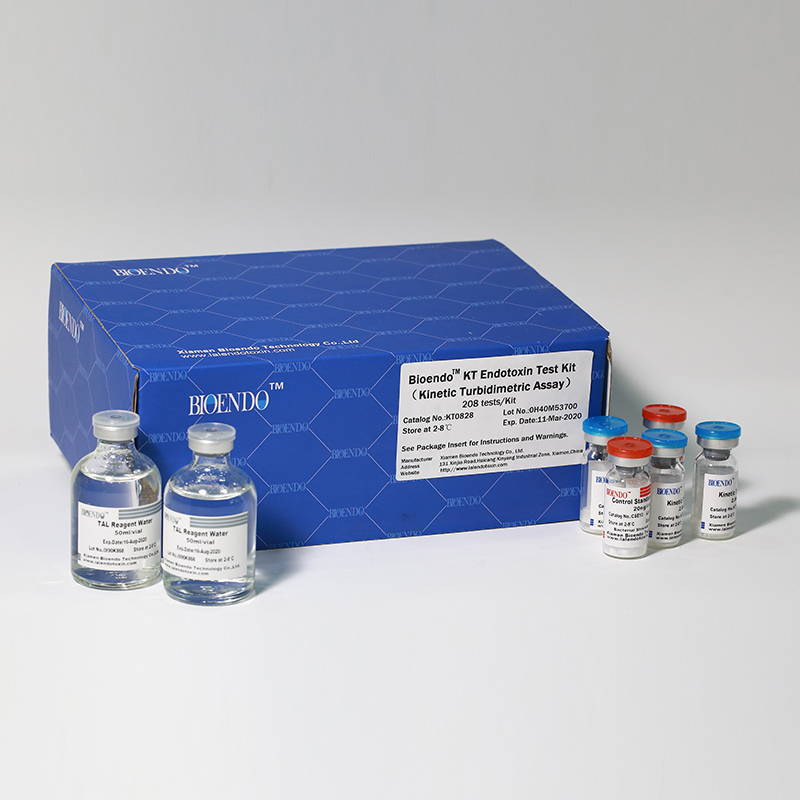
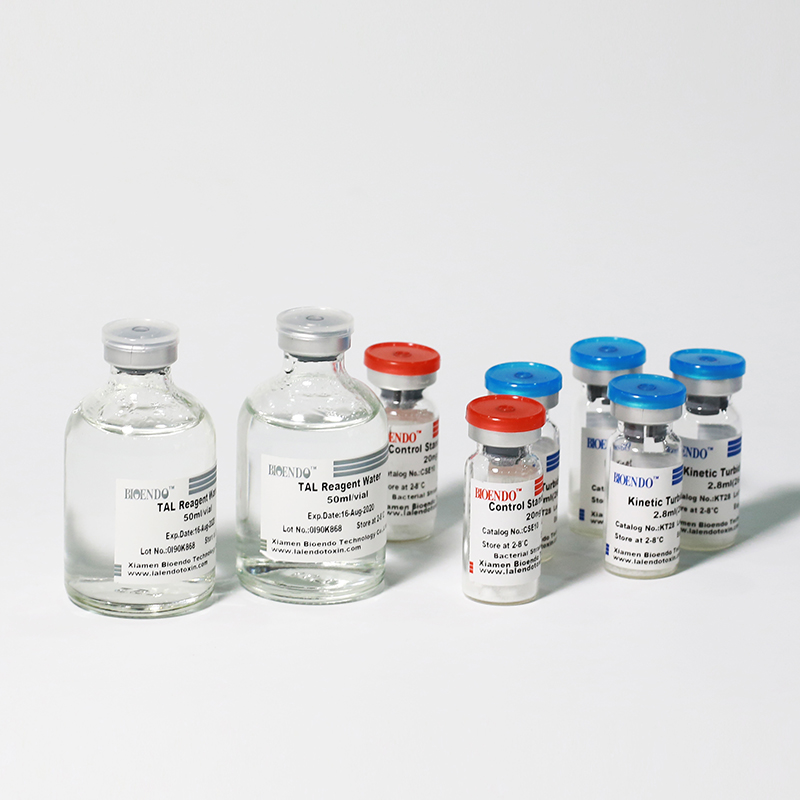

Related Product Guide:
We always continually offer you the most conscientious purchaser services, and the widest variety of designs and styles with finest materials. These efforts include the availability of customized designs with speed and dispatch for Low price for Bacterial Endotoxin Test Kinetic Turbidimetric Method – Kinetic Turbidimetric Amebocyte Lysate Vial – Bioendo , The product will supply to all over the world, such as: Singapore, Canada, South Korea, Our company now has many department, and there have more than 20 employees in our company. We set up sales shop, show room, and product warehouse. In the meantime, we registered our own brand. We have tightened inspection for quality of product.
This supplier stick to the principle of "Quality first, Honesty as base", it is absolutely to be trust.






