Hot-selling LAL Turbidimetric test - Kinetic Turbidimetric Amebocyte Lysate Vial – Bioendo
Hot-selling LAL Turbidimetric test - Kinetic Turbidimetric Amebocyte Lysate Vial – Bioendo Detail:
Kinetic Turbidimetric Amebocyte Lysate Vial
1. Product Introduction
Kinetic Turbidimetric Amebocyte Lysate Vial is developed based on the principle that the time needed to reach a certain absorbance increase (onset OD), i.e. onset time, is negatively correlated with the endotoxin concentration. Sensitivity could reach 0.005EU/ml, and the detection could reach four orders of magnitude. It is specially suitable for pharmaceuticals industry to monitor endotoxin concentration.
2. Product Parameter:
Assay range:0.005-50EU/ml; 0.01 – 10EU/ml
3. Product Application
End-product endotoxin (pyrogen) qualification, Water for injection endotoxin assay, raw material endotoxin testing or endotoxin level monitoring during manufacturing process for pharmaceutical companies or medical devices manufacturers.
Note:
Lyophilized Amebocyte Lysate reagent manufactured by Bioendo is made from amebocyte lysate from the horseshoe crab (Tachypleus tridentatus).
|
Catalog No. |
ml/vial |
Tests/Vial |
Vials/Pack |
Sensitivity EU/ml |
|
KT17 |
1.7 |
16 |
10 |
0.01-10EU/ml |
|
KT17S |
1.7 |
16 |
10 |
0.005-5EU/ml, 0.01-10EU/ml |
|
KT52 |
5.2 |
50 |
10 |
0.01-10EU/ml |
|
KT52S |
5.2 |
50 |
10 |
0.005-5EU/ml, 0.01-10EU/ml |
The Lyophilized Amebocyte Lysate reagent sensitivity and the Control Standard Endotoxin potency are assayedagainst USP Reference Standard Endotoxin. The Lyophilized Amebocyte Lysate reagent kits come withproduct instruction, Certificate of Analysis.
Product detail pictures:
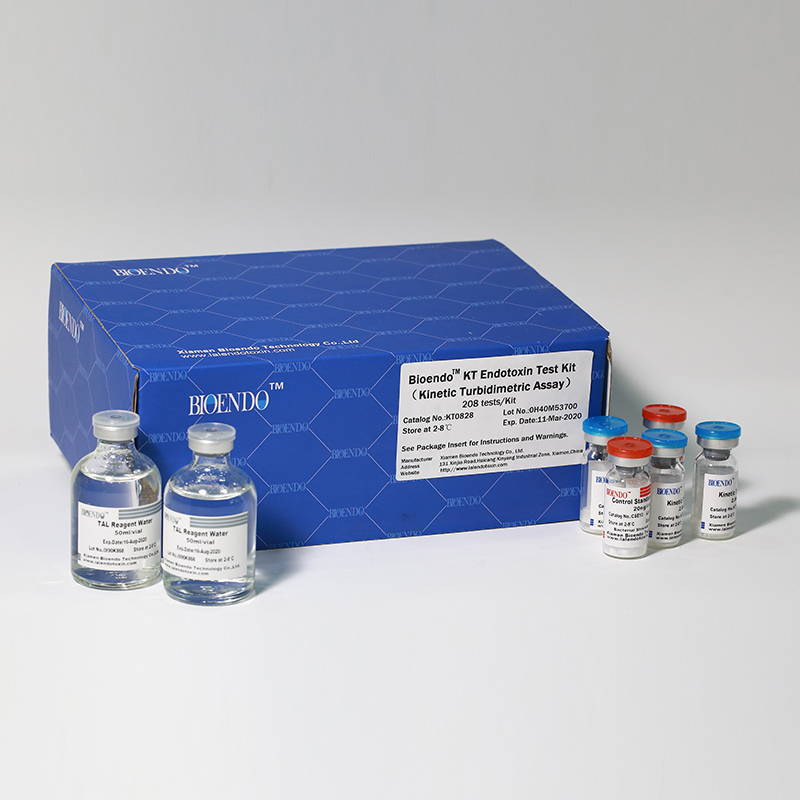
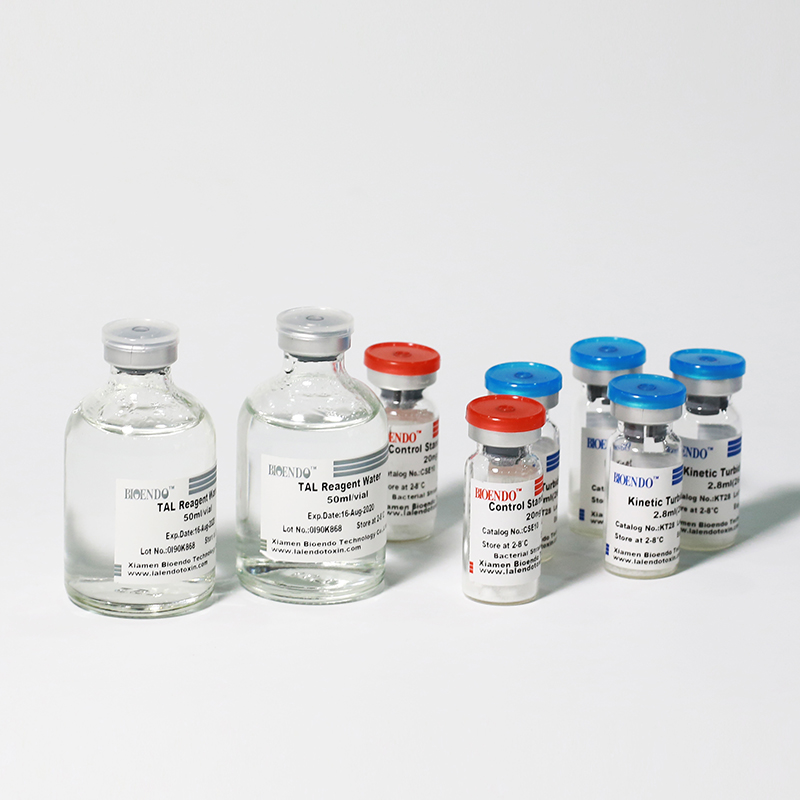

Related Product Guide:
We aim to find out quality disfigurement from the production and supply the best service to domestic and overseas customers wholeheartedly for Hot-selling LAL Turbidimetric test - Kinetic Turbidimetric Amebocyte Lysate Vial – Bioendo , The product will supply to all over the world, such as: Thailand, Somalia, Greece, We always adhere to follow the honesty, mutual benefit, common development, after years of development and the tireless efforts of all staff, now has perfect export system, diversified logistics solutions, comprehensive meet customer shipping, air transport, international express and logistics services. Elaborate one-stop sourcing platform for our customers!
Although we are a small company, we are also respected. Reliable quality, sincere service and good credit, we are honored to be able to work with you!






