Good Quality LAL Test - Single-Channel Mechanical Pipettor – Bioendo
Good Quality LAL Test - Single-Channel Mechanical Pipettor – Bioendo Detail:
Single-Channel Mechanical Pipettor
1. Product Information
Single channel mechanical pipette is ideal tool to support endotoxin detection with Lyophilized Amebocyte Lysate which covers gel-clot technique, kinetic turbidimetric technique, kinetic chromogenic technique, and end-point chromogenic technique. All pipettors are produced by following ISO8655 – 2:2002. The quality control involves gravimetric testing of each pipette with distilled water at 22℃.
2. Product Features:
- Light weight, economic, low force design
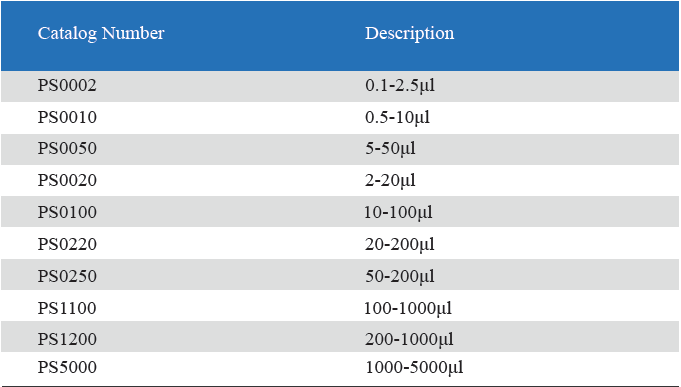
- Measuring volume range from 0.1μL to 5mL
- Easy to calibrate and maintain with tool supplied
- Design helps to avoid repetitive strain injuries
- Calibrated in accordance with ISO8655. Each pipettor supplied with individual test certificate
- The low part is available for autoclaving
Product detail pictures:

Related Product Guide:
As for aggressive costs, we believe that you will be searching far and wide for anything that can beat us. We can state with absolute certainty that for such high-quality at such rates we have been the lowest around for Good Quality LAL Test - Single-Channel Mechanical Pipettor – Bioendo , The product will supply to all over the world, such as: Madras, Seattle, Orlando, Since its foundation , the company keeps living up to the belief of "honest selling , best quality , people-orientation and benefits to customers. " We are doing everything to offer our customers with best services and best products . We promise that we'll be responsible all the way to the end once our services begin.
It's really lucky to find such a professional and responsible manufacturer, the product quality is good and delivery is timely, very nice.







