Factory source Advantage Of LAL Test - Single-Channel Mechanical Pipettes (Semi-sterile) – Bioendo
Factory source Advantage Of LAL Test - Single-Channel Mechanical Pipettes (Semi-sterile) – Bioendo Detail:
Single-Channel Mechanical Pipettes (Semi-sterile)
1. Product Information
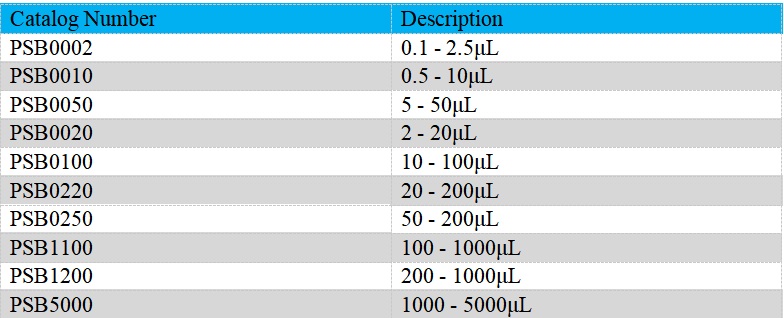
The Single-Channel Mechanical Pipettes (Semi-sterile) is adjustable, which could be used to dispense liquid precisely. Measuring volume of our Single-Channel Mechanical Pipettes (Semi-sterile) ranges from 0.1μL to 5mL. Products are produced based on ISO8655/DIN12650. It could be widely employed on endotoxin detection, etc..
2. Product Features
- Light weight, economic
- The pipettes cover volume range from 0.1μL to 5mL
- Semi-sterile
- Easy calibration and maintenance
Product detail pictures:

Related Product Guide:
It can be our accountability to satisfy your preferences and competently provide you. Your satisfaction is our greatest reward. We are searching ahead towards your visit for joint growth for Factory source Advantage Of LAL Test - Single-Channel Mechanical Pipettes (Semi-sterile) – Bioendo , The product will supply to all over the world, such as: Peru, Sri Lanka, Denmark, It is our customers' satisfaction over our products and services that always inspires us to do better in this business. We build mutually beneficial relationship with our clients by giving them large selection of premium car parts at marked down prices. We provide wholesale prices on all our quality parts so you are guaranteed greater savings.
The supplier abide the theory of "quality the basic, trust the first and management the advanced" so that they can ensure a reliable product quality and stable customers.








