Factory For Bacterial Endotoxin Test Usp Chapter - Control Standard Endotoxin (CSE) – Bioendo
Factory For Bacterial Endotoxin Test Usp Chapter - Control Standard Endotoxin (CSE) – Bioendo Detail:
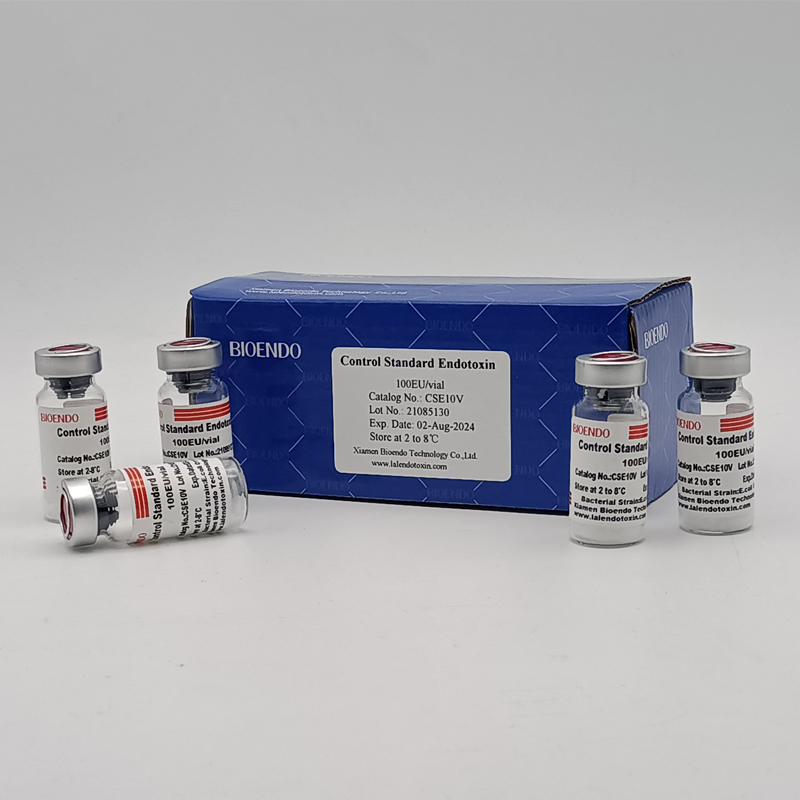
Control Standard Endotoxin (CSE)
1. Product Information
Control Standard Endotoxin (CSE) is extracted from E.coli O111:B4. CSE is an economic alternative to Reference Standard Endotoxin (RSE) in constructing standard curves, validating product and preparing controls in Lyophilized Amebocyte Lysate test. The labeled potency of CSE endotoxinE.coli standard is referenced against RSE. The Control Standard Endotoxin could be used with gel clot assay, kinetic turbidimetric assay or kinetic chromogenic assay as the endotoxin testing standards. The Certificate of Analysis will show the matched Lyophilized Amebocyte Lysate reagent lots.
2. Product Parameter
| Catalog Number | Potency (EU/vial) | Package |
| CSE10V | 100 to 999 EU | seal in glass vial, 10vials/pack |
| CSE100V | 1 to 199 EU | seal in glass vial, 10vials/pack |
| CSE10A | 1 to 99 EU | seal in glass ampoule, 10vials/pack |
3. Product Feature and Application
Bioendo CSE was labeled by the potency and matched to Lyophilized Amebocyte Lysate reagent lots. Users do not need to do the CSE/RSE ratio assay. Low potency control standard endotoxin is available to avoid lots of steps of dilution to provide convenience for end users.
Product Condition:
Control Standard Endotoxin (CSE), extracted from E.coli O111:B4, is an economic alternative to Reference Standard Endotoxin (RSE) in constructing standard curves, validating product and preparing controls in endotoxin test. The potency of CSE is referenced against USP Reference Standard Endotoxin, and labeled in the Certificate of Analysis.
Endotoxin test assay: Lysate reagent and CSE lot number have to be matched.
Pyrogen free tip box
Endotoxin free tubes
Product detail pictures:

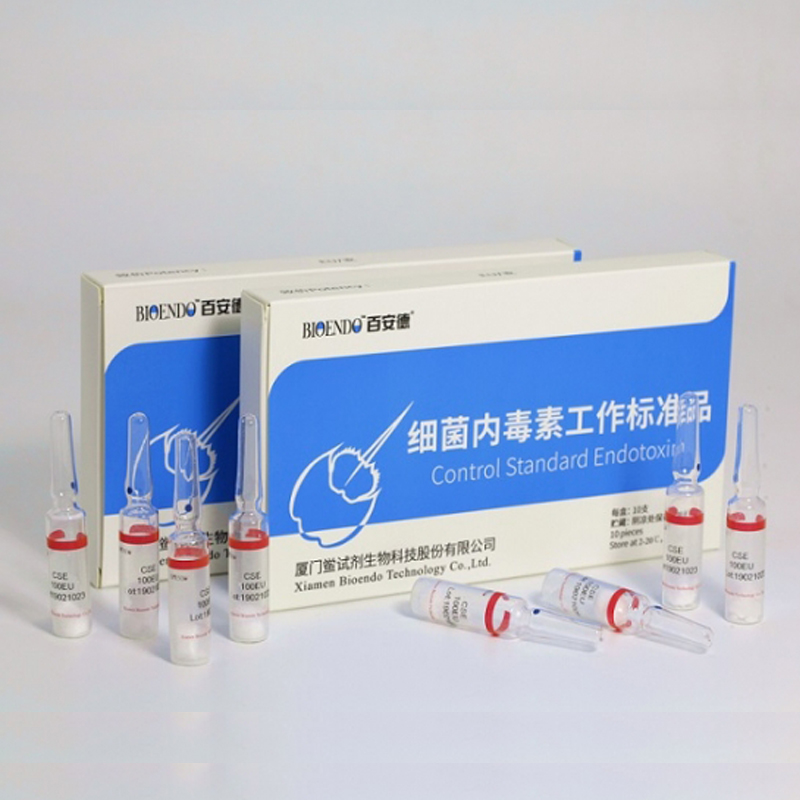

Related Product Guide:
abide by the contract", conforms into the market requirement, joins in the market competition by its good quality also as provides a lot more comprehensive and great company for purchasers to let them turn into huge winner. The pursue from the firm, would be the clients' gratification for Factory For Bacterial Endotoxin Test Usp Chapter - Control Standard Endotoxin (CSE) – Bioendo , The product will supply to all over the world, such as: Kuala Lumpur, Armenia, Namibia, Strong infrastructure is the need of any organization. We are backed with a robust infrastructural facility that enables us to manufacture, store, quality check and dispatch our products worldwide. To maintain smooth work flow, we have sectioned our infrastructure into a number of departments. All these departments are functional with latest tools, modernized machines and equipment. Owing to which, we are able to accomplish voluminous production without compromising upon the quality.
Company director has very rich management experience and strict attitude, sales staff are warm and cheerful, technical staff are professional and responsible,so we have no worry about product,a nice manufacturer.