Factory Cheap Hot endotoxin removal kit – EtEraser™ SE Endotoxin Removal Kit – Bioendo
Factory Cheap Hot endotoxin removal kit – EtEraser™ SE Endotoxin Removal Kit – Bioendo Detail:
EtEraser™ SE Endotoxin Removal Kit
1. Product information
Lipopolysaccharide (LPS) is a bacterial endotoxin and a majorconstituent of the cell walls of gramnegative bacteria. Recombinant proteinfrom E.coli usually contains high level of endotoxins. The removal of theseendotoxins is highly necessary for downstream processes.
EtEraser SE Endotoxin Removal kit are designedto remove endotoxin contamination from aqueous solutions. The kits containendotoxin removal resin binds to reduce endotoxin levels in protein samples by≥99% in less than 2 hours.
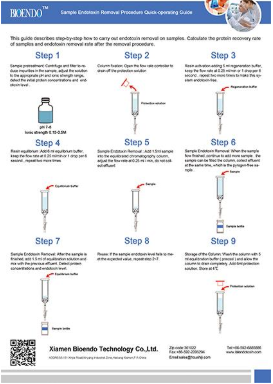
The kit includes a pre-packed endotoxin removal column1.5 ml, Equilibration Buffer, Regeneration Buffer and pyrogen-freecollection tube and tips. The column has a high binding capacity of > 2,000, 000 EU / ml. This product may be reused up to five times if properlyregenerated. The affinity resin is available in slurry and could be up-scalingin biopharmaceutical process.
EtEraser SE Endotoxin Removal kit utilizes food grade modified ε-poly-L-lysine ashigh affinity ligand for endotoxins. The resin is agarose beads which is verystable and has no cytotoxic effect to human body. After purification, therewill no toxic residual remained in the samples. This endotoxin removal kit canbe used in protein drugs, vaccines, antibodies, DNA/RNA, polysaccharide andother biological samples.
2. Product features
• high stability — does not affect the activity of most of thebiological sample
• high bonding capacity — >2000000 EU/ml
• non-toxic —–> Non-toxic food grade ligands, no cytotoxiceffects poly lysine
• high protein recovery — >95% protein recovery for proteinsamples
• high removal efficiency — remove >99% endotoxin,endotoxin level in the sample can be less than 0.1 EU/ml after endotoxinremoving process
• wide application range — can be used in endotoxin removalfor proteins, peptides, antibodies, vaccines, polysaccharide and otherbiological samples


Product detail pictures:

Related Product Guide:
"Sincerity, Innovation, Rigorousness, and Efficiency" is the persistent conception of our firm to the long-term to develop together with consumers for mutual reciprocity and mutual advantage for Factory Cheap Hot endotoxin removal kit – EtEraser™ SE Endotoxin Removal Kit – Bioendo , The product will supply to all over the world, such as: Paraguay, Liberia, Doha, We integrate all our advantages to continuously innovate, improve and optimize our industrial structure and product performance. We will always believe in and work on it. Welcome to join us to promote green light, together we will make a better Future!
The company can keep up with the changes in this industry market, product updates fast and the price is cheap, this is our second cooperation, it's good.