8 Year Exporter Define LAL Test - Single-Channel Mechanical Pipettor – Bioendo
8 Year Exporter Define LAL Test - Single-Channel Mechanical Pipettor – Bioendo Detail:
Single-Channel Mechanical Pipettor
1. Product Information
Single channel mechanical pipette is ideal tool to support endotoxin detection with Lyophilized Amebocyte Lysate which covers gel-clot technique, kinetic turbidimetric technique, kinetic chromogenic technique, and end-point chromogenic technique. All pipettors are produced by following ISO8655 – 2:2002. The quality control involves gravimetric testing of each pipette with distilled water at 22℃.
2. Product Features:
- Light weight, economic, low force design
- Measuring volume range from 0.1μL to 5mL
- Easy to calibrate and maintain with tool supplied
- Design helps to avoid repetitive strain injuries
- Calibrated in accordance with ISO8655. Each pipettor supplied with individual test certificate
- The low part is available for autoclaving
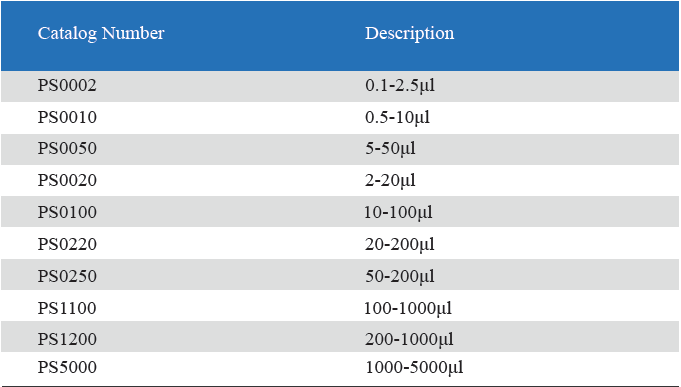
Product detail pictures:

Related Product Guide:
Bear "Customer first, Quality first" in mind, we work closely with our customers and provide them with efficient and professional services for 8 Year Exporter Define LAL Test - Single-Channel Mechanical Pipettor – Bioendo , The product will supply to all over the world, such as: Kyrgyzstan, Jakarta, Greece, Adhering to the principle of "Enterprising and Truth-Seeking, Preciseness and Unity", with technology as the core, our company continues to innovate, dedicated to providing you with the highest cost-effective solutions and meticulous after-sales service. We firmly believe that: we're outstanding as we have been specialized.
This supplier stick to the principle of "Quality first, Honesty as base", it is absolutely to be trust.