2022 China New Design limulus amebocyte lysate price - Endotoxin-specific Lyophilized Amebocyte Lysate – Bioendo
2022 China New Design limulus amebocyte lysate price - Endotoxin-specific Lyophilized Amebocyte Lysate – Bioendo Detail:
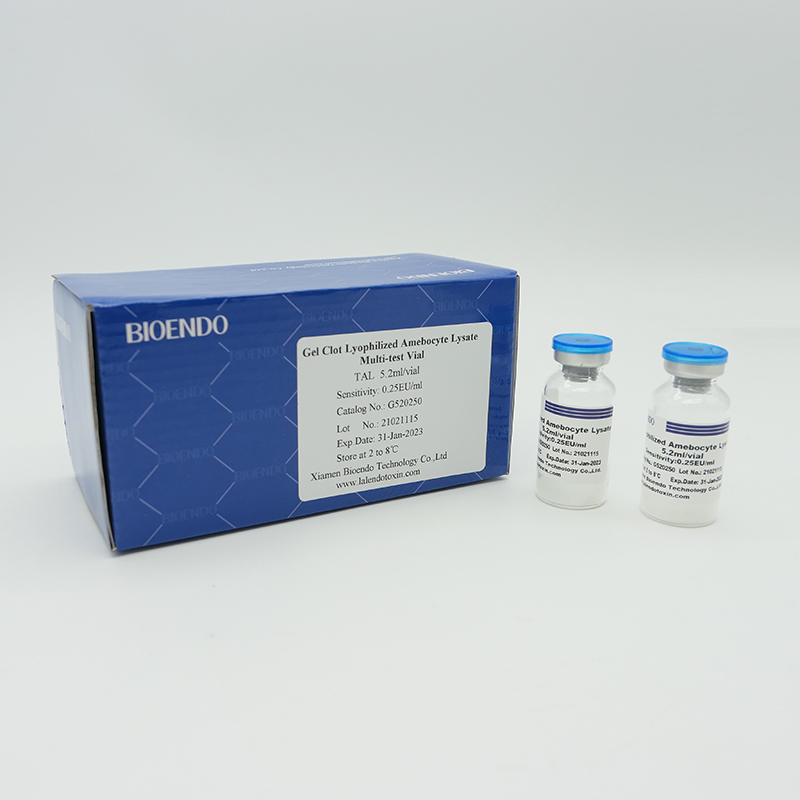
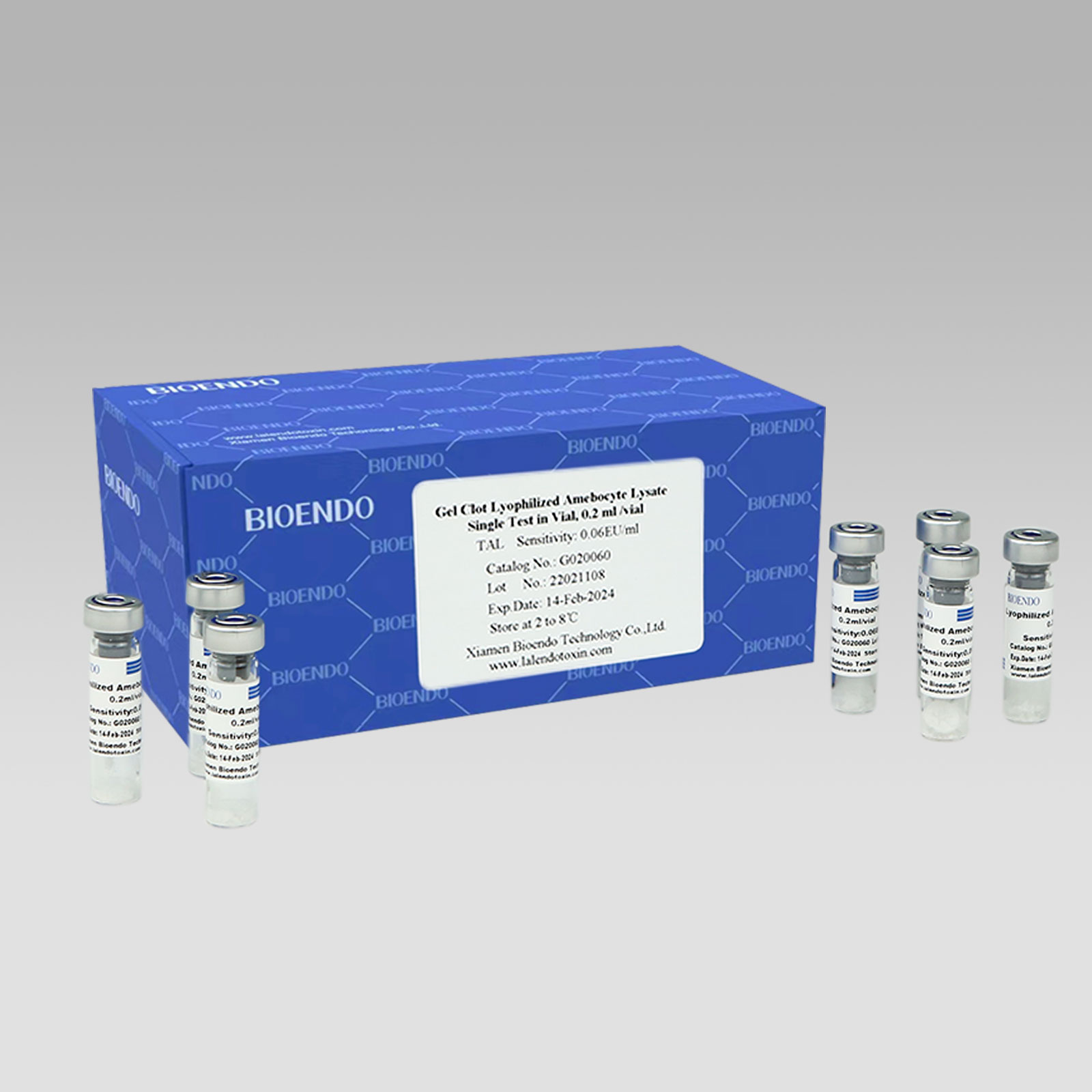
Endotoxin-specific Lyophilized Amebocyte Lysate
1. Product Information
Lyophilized Amebocyte Lysate extracted from horseshoe crab contains factor C pathway which reacts to endotoxin and factor G pathway which reacts to (1,3)- ß-D-Glucan. Endotoxin-specific Lyophilized Amebocyte Lysate contains ß-Glucan inhibitor, which will avoid false positive, result happening between factor C pathway and (1,3)- ß-D-Glucan. Endotoxin-specific Lyophilized Amebocyte Lysate is especially suitable for samples with potential which are contaminated by ß-Glucan. Bioendo has two Endotoxin-specific Lyophilized Amebocyte Lysate. One is that operator needs to add 0.1ml Water for BET and 0.1ml sample solution to the ampoule of Endotoxin-specific Lyophilized Amebocyte Lysate respectively; the other is that 0.2ml sample solution could be added directly into the ampoule.
2. Product Parameter
Sensitivity range: 0.03, 0.06, 0.125, 0.25, 0.5EU/ml
3. Product Application
Endotoxin-specific Lyophilized Amebocyte Lysate is the optimal choice for endotoxin detection of samples contaminated by beta-glucan such as cellulose, fungi solution.
Note:
Lyophilized Amebocyte Lysate (LAL) reagent manufactured by Bioendo is made from amebocyte lysate derived blood of horseshoe crab.
|
Catalog No. |
Sensitivity (EU/ml) |
|
ES010030 |
0.03 |
|
ES010060 |
0.06 |
|
ES010125 |
0.125 |
|
ES010250 |
0.25 |
|
ES010500 |
0.5 |
Product Condition:
The sensitivity of Lyophilized Amebocyte Lysate and Control Standard Endotoxin potency are assayed against USP Reference Standard Endotoxin. Lyophilized Amebocyte Lysate kit comes with product instruction, Certificate of Analysis, MSDS.
Lysate reagent + Matched Control standard endotoxin ( save cost than RSE, CSE vial is calibrated by RSE. )
BET water or called LAL reagent water
Endotoxin free tubes and tips
Above all materials are necessary in the operation of endotoxin test assay.
Product detail pictures:


Related Product Guide:
As a result of ours specialty and service consciousness, our company has won a good reputation among customers all over the world for 2022 China New Design limulus amebocyte lysate price - Endotoxin-specific Lyophilized Amebocyte Lysate – Bioendo , The product will supply to all over the world, such as: Ecuador, Islamabad, Madagascar, We take measure at any price to attain essentially the most up-to-date gear and procedures. The packing of nominated brand is our a further distinguishing feature. The solutions to assure years of trouble-free service has attracted a great deal customers. The goods are obtainable in improved designs and richer variety, they're produced scientifically of purely raw supplies. It accessible in a variety of designs and specifications for the selection. The newest forms are much far better than the previous one and they're extremely popular with several clients.
The supplier abide the theory of "quality the basic, trust the first and management the advanced" so that they can ensure a reliable product quality and stable customers.