2022 China New Design LAL water testing - Single-Channel Mechanical Pipettor – Bioendo
2022 China New Design LAL water testing - Single-Channel Mechanical Pipettor – Bioendo Detail:
Single-Channel Mechanical Pipettor
1. Product Information
Single channel mechanical pipette is ideal tool to support endotoxin detection with Lyophilized Amebocyte Lysate which covers gel-clot technique, kinetic turbidimetric technique, kinetic chromogenic technique, and end-point chromogenic technique. All pipettors are produced by following ISO8655 – 2:2002. The quality control involves gravimetric testing of each pipette with distilled water at 22℃.
2. Product Features:
- Light weight, economic, low force design
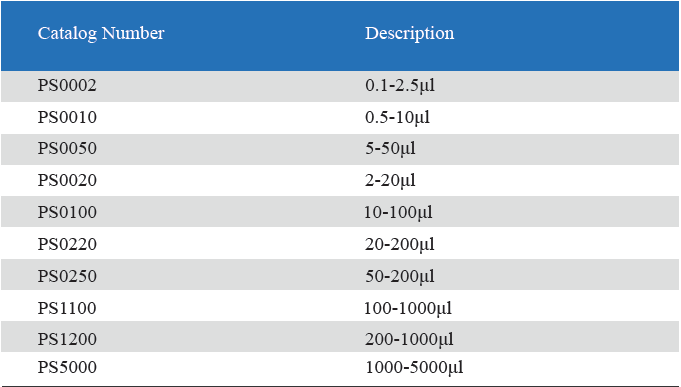
- Measuring volume range from 0.1μL to 5mL
- Easy to calibrate and maintain with tool supplied
- Design helps to avoid repetitive strain injuries
- Calibrated in accordance with ISO8655. Each pipettor supplied with individual test certificate
- The low part is available for autoclaving
Product detail pictures:

Related Product Guide:
Fast and superior quotations, informed advisers to help you choose the correct merchandise that suits all your requirements, a short generation time, responsible quality control and different services for paying and shipping affairs for 2022 China New Design LAL water testing - Single-Channel Mechanical Pipettor – Bioendo , The product will supply to all over the world, such as: luzern, Malta, Russia, Corporate goal: Customers' satisfaction is our goal, and sincerely hope to establish long-terms stable cooperative relations with customers to jointly develop the market. Building brilliant tomorrow together!Our company regards "reasonable prices, efficient production time and good after-sales service" as our tenet. We hope to cooperate with more customers for mutual development and benefits. We welcome potential buyers to contact us.
The company comply with the contract strict, a very reputable manufacturers, worthy a long-term cooperation.